ABSTRACT
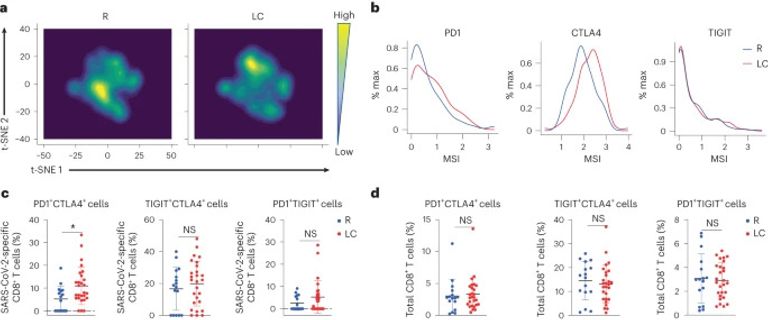
Long COVID (LC) occurs after at least 10% of severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) infections, yet its etiology remains poorly understood. We used ‘omic” assays and serology to deeply characterize the global and SARS-CoV-2-specific immunity in the blood of individuals with clear LC and non-LC clinical trajectories, 8 months postinfection. We found that LC individuals exhibited systemic inflammation and immune dysregulation. This was evidenced by global differences in T cell subset distribution implying ongoing immune responses, as well as by sex-specific perturbations in cytolytic subsets. LC individuals displayed increased frequencies of CD4+ T cells poised to migrate to inflamed tissues and exhausted SARS-CoV-2-specific CD8+ T cells, higher levels of SARS-CoV-2 antibodies and a mis-coordination between their SARS-CoV-2-specific T and B cell responses. Our analysis suggested an improper crosstalk between the cellular and humoral adaptive immunity in LC, which can lead to immune dysregulation, inflammation and clinical symptoms associated with this debilitating condition.